“Improvement of sperm quality after micronutrient supplementation”
Open Comparative Study 2010
Study design
Headed by Univ.-Prof. Dr. Martin Imhof, this open comparative study was carried out at the Fertility Centre at the Vienna International Medical Clinic in Austria between January 2006 and October 2010. A group of 132 sub-fertile men (treatment arm) was invited to take two daily capsules of the active ingredient (PROfertil®) for a period of three months between the first and subsequent sperm analysis. Each capsule with the active ingredient contained L-carnitine, L-arginine, zinc, vitamin E, glutathione, folic acid, selenium and coenzyme Q10. The control group consisted of sub-fertile men who were not given the active ingredient (n = 73). The relevant target measurement method was standardized sperm analysis, carried out at the beginning and after three months of treatment. The inclusion criteria were age >18 and < 65 years, infertility for a year and at least two abnormal semen analyses (also called spermiogram or sperm test) prior to the study. Exclusion criteria were varicose veins, infections, aspermia or hormonal disorders.
Study results in detail:
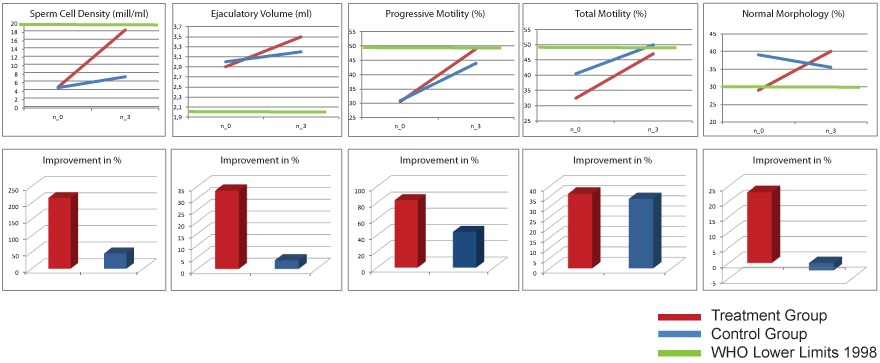
Improvement of sperm quality in sub-fertile men due to the micronutrient combination 23,24
All evaluated parameters of the sperm analysis were significantly higher after three months of treatment with the active ingredient. Sperm density, average ejaculate volume, sperm motility (forward moving and total) and normal morphology increased by 33.3, 215.5, 83.1, 36.4 and 23.0% respectively. These improvements were significantly higher than those in the control group. No side effects have been recorded in the treatment group and after a six-month observation period 34 pregnancies were reported, compared with only 11 pregnancies in the control group.
Conclusion:
The study showed impressively that the administration by PROfertil® achieves a significant improvement in sperm quality up to a completely normal semen analysis.